COVID-19 I (2020): Policies, Risks, Preparedness
SciOPS conducted the initial COVID-19 survey of academic scientists in May 2020 to look at how COVID-19 policies have influenced academic scientists’ home-life, research, and views on policy.
Question
Finding
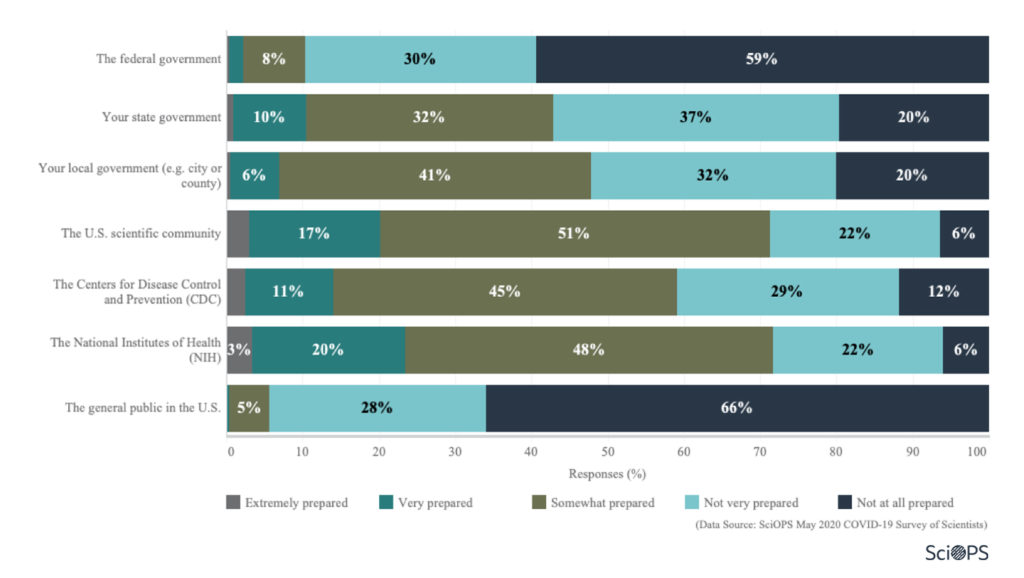
About 90% of responding scientists believe the federal government was not very prepared or not prepared at all for the COVID-19 disease pandemic.
Scientists believe that state (43%) and local (48%) governments were at least somewhat prepared.
Though scientists are more positive about the preparedness of the U.S scientific community, CDC and NIH, most believe the US general public was not very prepared or not prepared at all (94%).
Question
Finding
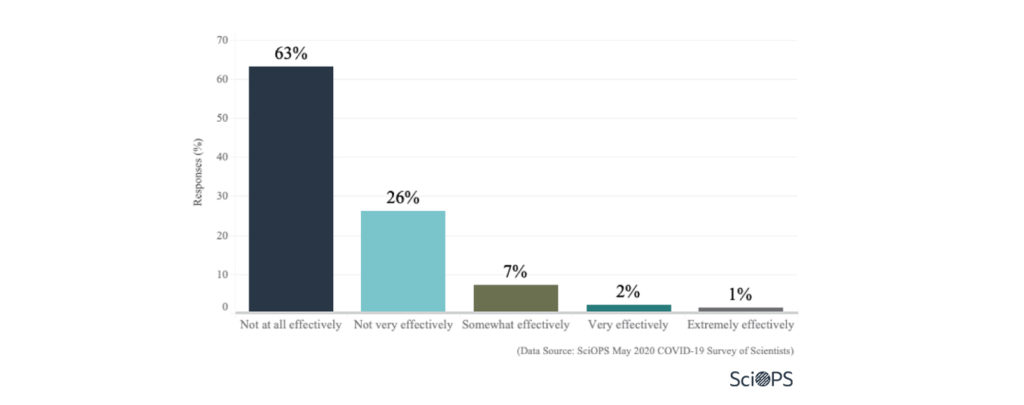
Most of the scientists (about 89%) said that the US government is not at all or not very effectively managing the COVID-19 pandemic.
Question
Using the COVID-19 disease as an example, what do you believe is the risk/benefit tradeoff associated with each of the following potential decisions that might need to be made during the time of a national pandemic emergency? (N=351)
Finding
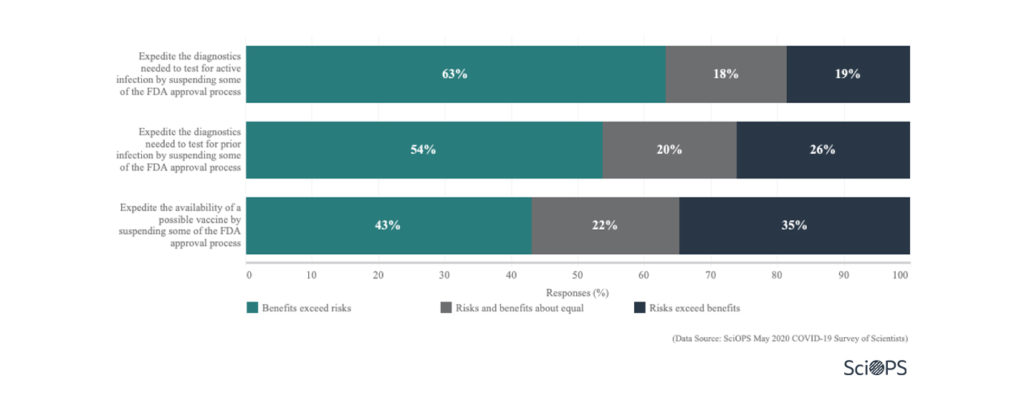
Scientists responded that the benefits exceed the risks when some of the FDA approval process are suspended:
- to expedite the diagnostics needed to test for active infection (About 63%).
- to expedite the diagnostics needed to test for prior infection ( About 54%).
- to expedite the availability of potential vaccine (About 43%).
Question
Finding
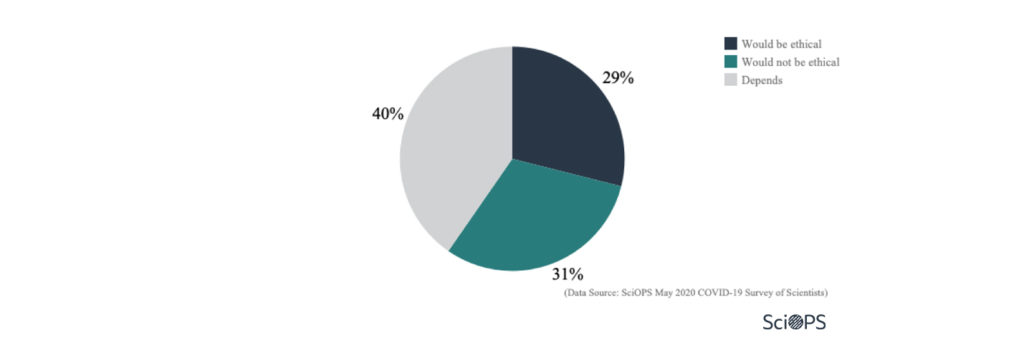
Around 30% of the scientists responded that it would be unethical to bypass some of the formal approval process to expedite the distribution of new COVID-19 vaccines.
Yet the largest number of scientists (about 40%) responded that the issue of ethics for bypassing some formal process to accelerate the vaccine distribution would depend on the situation.
Question
How necessary do you feel the use of such technologies are for mitigating a public health crisis such as the COVID-19 pandemic? (N=356)
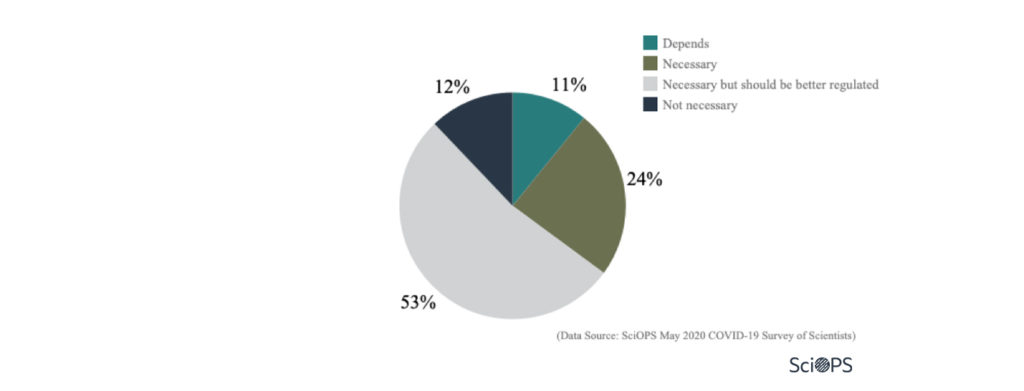
Finding
More than half of the scientists (53%) said that the use of technology for better surveillance is necessary, but needs better regulations.
About 24% of scientists indicated that the use of technology to handle the public health crisis is necessary.
Related Publications
A more in depth analysis of this study can be found in our recent publication “COVID-19 and the academy: opinions and experiences of university-based scientists in the U.S.” in Humanities and Social Sciences Communications.
You can also read policy memos about this study and others on Arizona State University’s Center for Science, Technology and Environmental Policy Studies website.
Survey Description
This national survey of scientists and engineers on the impacts of COVID-19 on academic research was conducted by the Center for Science, Technology and Environmental Policy Studies at Arizona State University. The project seeks to understand how COVID-19 and related policies impact academic research and funding, issues related to COVID-19 science communication, and regulation of COVID-19 research. The survey collected data on individual background and perceptions of impacts, behavior and expectations due to COVID-19. The population of the survey included tenured and tenure-track academic scientists from three fields (biology, civil and environmental engineering, and biochemistry) at 21 randomly selected universities classified as Carnegie designated research extensive (R1) universities in the United States.
The online survey was administered in English using Sawtooth Software®. 1,968 individuals were invited to participate in the survey via email with a series of personalized email follow-ups. The survey was opened on May 7 and closed on May 29, 2020. It resulted in 373 usable responses, with a response rate of 21% (AAPOR RR4). Responses are mostly from male respondents (63.5%), which is only slightly lower than the gender distribution in the population. The distribution of field also reflects the sampling frame, with 68.9% of respondents working in biology and genetics departments, 9.4% working in biochemistry departments and 21.7% working in civil and environmental engineering departments. The survey sample was weighted by gender and academic field. The measure of sampling error for questions answered by the full sample is plus or minus 5 percentage points.
